The Lunula Laser is the easy and convenient treatment that helps turn your discolored and disfigured nails into clearer, healthier looking nails. 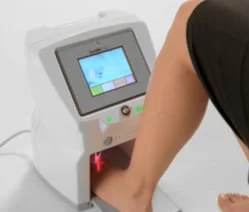
- The only touchless, painless treatment for onychomycosis
- No pain, no downtime, and no risk
- No more embarrassment about unsightly feet
The Lunula Laser is the simple and painless solution to effectively treat onychomycosis. The Lunula Laser uses non-thermal low-level laser light to treat foot fungus and requires very little time or set-up. It’s the only system you can turn on and walk away—the device is already pre-set.
From baseline to 6 months post treatment patients experienced 6.15mm of new clear nail growth or 37% clearing of the nail. The following before and after photos were part of Erchonia's clinical trials after 4 treatments:
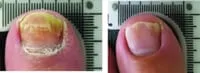
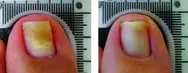
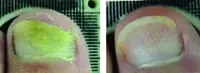
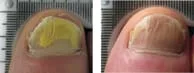
Toenail fungus is a condition that many people develop and suffer from. Toenail fungus often results in the discoloration and thickening of the toenail, which can cause embarrassment for many people. The condition begins as a small white or yellow growth beneath the tip of the nail and at its worse can develop into the nail’s deterioration.
Thick, ragged, and distorted nails lacking shine are a sign of toenail fungus. The color of the affected nail tends to be darker because of debris buildup underneath the nail. Toenail fungus is more likely to develop for people who wear socks and shoes that do not allow decent ventilation of the feet. Public showers, gyms, and swimming pools are breeding grounds for toenail fungus, as their damp and humid environments allow for the growth of bacteria.
Patients should make an appointment if they suspect they have toenail fungus because in its more severe stages, toenail fungus can become very painful. Other infections can develop from toenail fungus that can spread to other parts of the body.
Click here for more information on the Lunula Laser .
